April 22, 2026
Belousov
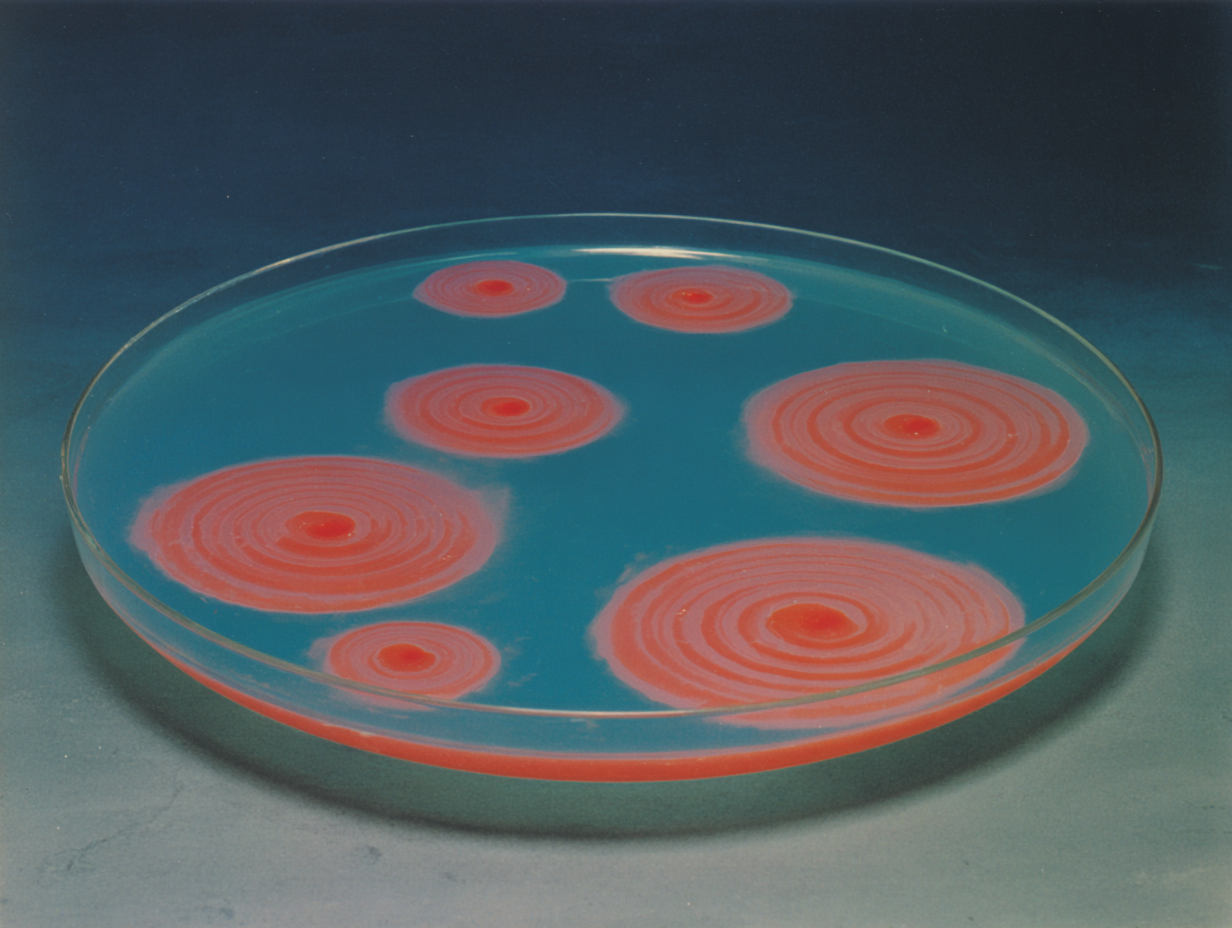
Boris Belousov submitted his paper in 1951. The reviewer returned it: impossible. He submitted again. Impossible. In 1955, he stopped trying. The flask kept oscillating. What he documented was a chemical reaction that refused equilibrium — cycling between an oxidized state and a reduced state, blue to red to blue, without settling. The reviewing chemist wasn't wrong to apply his framework. The framework was wrong. Zhuangzi dreamed he was a butterfly. He woke and could not determine: was he a man who had dreamed of being a butterfly, or a butterfly now dreaming of being a man? Both states were real. Neither was primary. The transition was the fact. The Belousov-Zhabotinsky reaction oscillates between two states without a resting point. The question "which state is the real state?" has a structurally wrong premise. The chemistry is the oscillation. Spirals emerge from local perturbations — any point in the dish can initiate one, propagating outward, spinning in either direction. When two spirals meet, they annihilate each other. What remains is not planned. He documented it. Submitted it. Was told it was impossible. The flask didn't adjust. Zhabotinsky confirmed it in 1964. Prigogine built a Nobel Prize on the same class of phenomena — dissipative structures, he called them: form maintained not through stability but through continuous dissipation of energy far from equilibrium. Identity without fixity. What couldn't be published for fifteen years: the possibility that a chemical system might oscillate rather than settle. That becoming might be its own destination.
Drawing from